Cartiva® Interposition Arthroplasty
Cartiva® interposition is an FDA-approved synthetic cartilage implant used to improve pain and function in individuals with a condition called hallux rigidus or stiff big toe caused by osteoarthritis of the first metatarsophalangeal (MTP) joint.
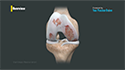
Osteoarthritis is a degenerative joint disease that causes breakdown and eventual loss of cartilage in the joint, causing a painful rubbing of bone surfaces with movement.
Cartiva® interposition arthroplasty is employed as a treatment for big toe osteoarthritis/hallux rigidus in which the damaged articular cartilage surface of the big toe is removed and replaced with a new Cartiva® interposition implant. The Cartiva® synthetic interposition implant is made of a durable, biocompatible, and slippery organic polymer called polyvinyl alcohol (PVA) that mimics the properties of natural cartilage and helps to relieve pain and restore normal range of motion and function to the big toe and foot.
Indications
Cartiva® interposition arthroplasty is indicated as a treatment option for hallux rigidus/osteoarthritis of the big toe when conservative treatment measures such as medications and physical therapy have failed to relieve symptoms such as:
- Pain and stiffness of the big toe
- Swollen and inflamed toe
- Damaged or worn-out cartilage
- Formation of bone spurs on the big toe joint
- Non-flexion of the big toe
- Narrowing of joint space
- Rubbing of the raw bone ends
- Foot pain that interferes with daily activities
Preparation
The pre-procedure preparation for Cartiva® interposition arthroplasty may involve the following:
- A review of your medical history and a physical examination
- Routine blood work and imaging
- Informing your doctor about medications or supplements you are taking
- Informing your doctor about any allergies to medications, anaesthesia, or latex
- Disclosing any recent illnesses or other medical conditions
- Refraining from medications such as blood thinners and nonsteroidal anti-inflammatories, or other supplements, if contraindicated for the procedure
- Arranging for someone to drive you home after the procedure
- Signing an informed consent form
Surgical procedure
Cartiva® interposition arthroplasty surgery is usually performed under general anaesthesia, and normally involves the following steps:
- You will be placed in a supine (face-up) position on the procedure table with your foot held in an optimal position to facilitate the surgery.
- An incision is made over the big toe, and soft tissues are moved away to expose the joint.
- Using special instruments, your surgeon will carefully remove the damaged portions of the joint, and a small hole – about 1 centimeter - is drilled into the head of the first metatarsal - the long bone connecting the mid-foot to the big toe joint.
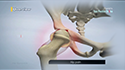
- Next, the Cartiva® interposition implant is placed into the hole. The synthetic implant is a tiny cylindrical plug with physical properties similar to those of articular cartilage.
- The implant provides a smooth, slippery, load-bearing surface to the joint, allowing it to move freely, relieving pain and stiffness, and improving joint function.
- Upon completion, the incision is closed with sutures and covered with a sterile bandage.
Postoperative care
In general, postoperative care instructions and recovery after Cartiva® interposition arthroplasty will involve the following:
- You may notice pain, swelling, and discomfort in the foot area. Pain and anti-inflammatory medications are provided as needed to address these.
- You are advised to keep your foot elevated as much as possible while resting to reduce swelling and pain.
- Applying ice packs to the foot several times a day is encouraged to reduce swelling and pain once medications are stopped.
- You should bend your knee, ankle, and toes periodically to stimulate circulation.
- Your big toe will be secured with a surgical shoe/sandal to facilitate wound healing, with instructions on restricted weight-bearing.
- Instructions on surgical site care and bathing will be provided to keep the surgical site clean and dry.
- Refrain from strenuous activities such as running and lifting heavy weights for a defined period.
- Most patients are able to resume their normal activities in a month or two after surgery, but with certain activity restrictions.
- Refrain from driving until you are fully fit and receive your doctor’s consent.
- Periodic follow-up appointments will be scheduled during which you will have your stitches removed, and you will be advised to gradually increase your activity level and gently exercise your big toe.
Risks and Complications
Cartiva® interposition arthroplasty is a relatively safe procedure; however, as with any surgery, some risks and complications may occur, such as the following:
- Damage to surrounding tissue
- Pain and discomfort
- Joint stiffness
- Infection
- Numbness
- Swelling and inflammation
- Deep vein thrombosis (DVT) or blood clots
Summary
The Cartiva® synthetic cartilage implant is a joint-preserving alternative to fusion surgery and is equally effective in relieving pain and restoring joint motion in patients with big toe osteoarthritis/hallux rigidus. It has a high success rate and most patients report 100% pain relief after treatment with Cartiva® interposition arthroplasty.